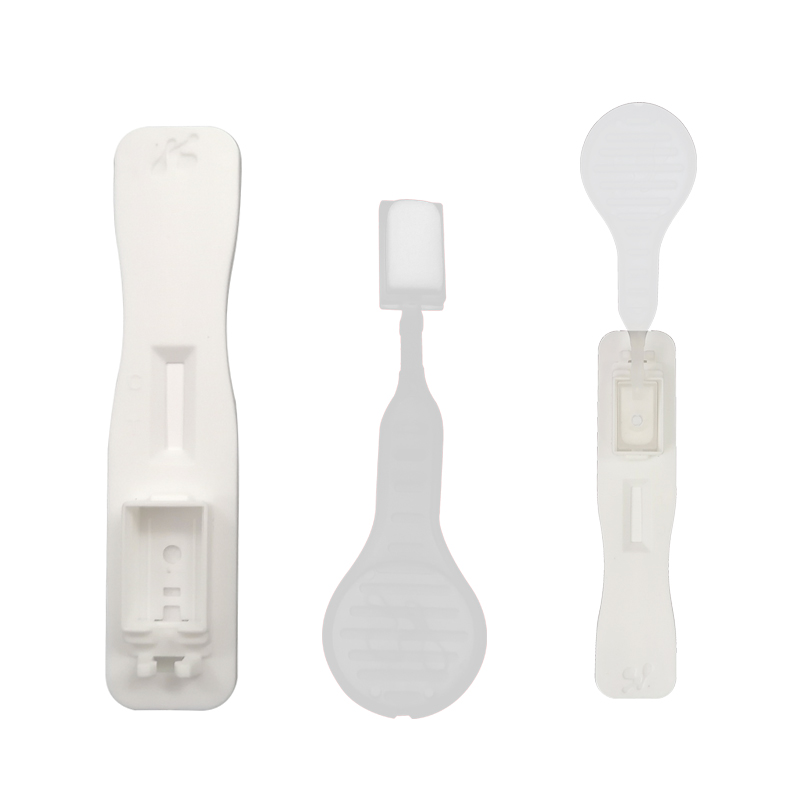
Lollipop saliva test (ICOVS-702G-1) rapid test strip plastic disposable rapid medical diagnosis antigen saliva test for 1 person
Intend Use:
◆Early screening and diagnosis applied for large-scale rapid screening in primary medical care.
◆COVID-19 Salivary Antigen Rapid Test Kit (Colloidal Gold) is an immunochromatographic assay for rapid, qualitative detection of severe acute respiratory syndrome coronavirus (SARS-CoV-2) Antigen from the saliva specimens.
◆The test provides preliminary test results. The test is to be used as an aid in the diagnosis of coronavirus infection disease (COVID-19), which is caused by SARS-CoV-2.
◆This product cannot be used as the basis to diagnose or exclude SARS-CoV-2 infection.
Sampling method
◆Saliva
Working Principle:
◆COVID-19 Salivary Antigen Rapid Test Kit (Colloidal Gold) is based on the principle of capture immunoassay for determination of SARS- CoV-2 Antigen from the saliva specimens.
◆When the specimen is added into the test device, the specimen is absorbed into the device by capillary action. If the sample contain novel coronavirus antigen, the antigen combined with the colloidal gold labeled novel coronavirus antibody, and when the novel coronavirus antigen level in the specimen is at or above the target cut-off, and the immune complex further binds to the coated antigen in the T line and this produces a colored test band that indicates a positive result.
◆When the novel coronavirus Antigen level in the specimen is zero or below the target cutoff, there is not a visible colored band in the Test Region of the device. This indicates a negative result.
◆To serve as a procedure control, a colored line will appear at the Control Region (C), if the test has been performed properly.
Product detail:
◆The novel coronaviruses belong to the β genus. COVID-19 is an acute respiratory infectious disease. People are generally susceptible. Currently, the patients infected by the novel coronavirus are the main source of infection; asymptomatic infected people can also be an infectious source. Based on the current epidemiological investigation, the incubation period is 1 to 14 days, mostly 3 to 7 days. The main manifestations include fever, fatigue, and dry cough. Nasal congestion, runny nose, sore throat, myalgia, and diarrhea are found in a few cases.
◆ Without Nasopharyngeal Swab
◆To be able to obtain the result rapidly within 15 mints
◆The procedure is simple and easy to operate
◆Specificity and Accuracy more than 99% and Sensitivity more then 96.3%
◆Europe registered in Germany, Italy and Spain and so on area.
How to use: